PCV2 DNA extracts from lungs and lymph nodes samples (from 2 wasting pigs) were sequenced for PCV2 genotyping.
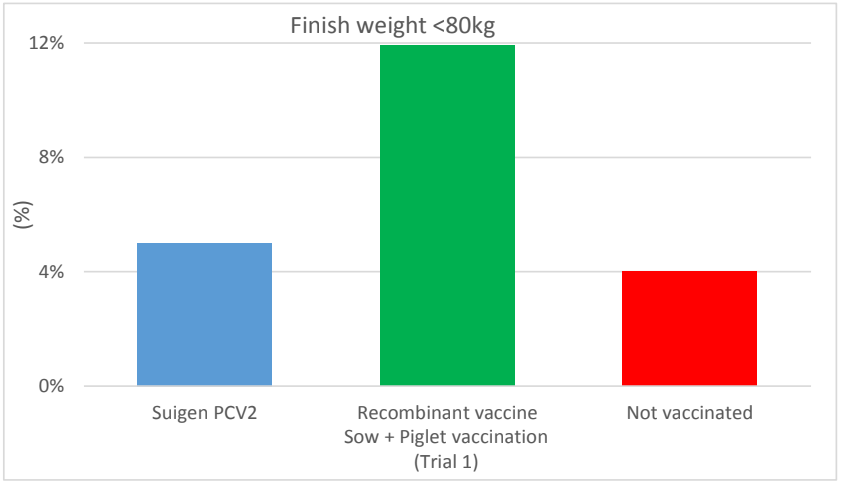
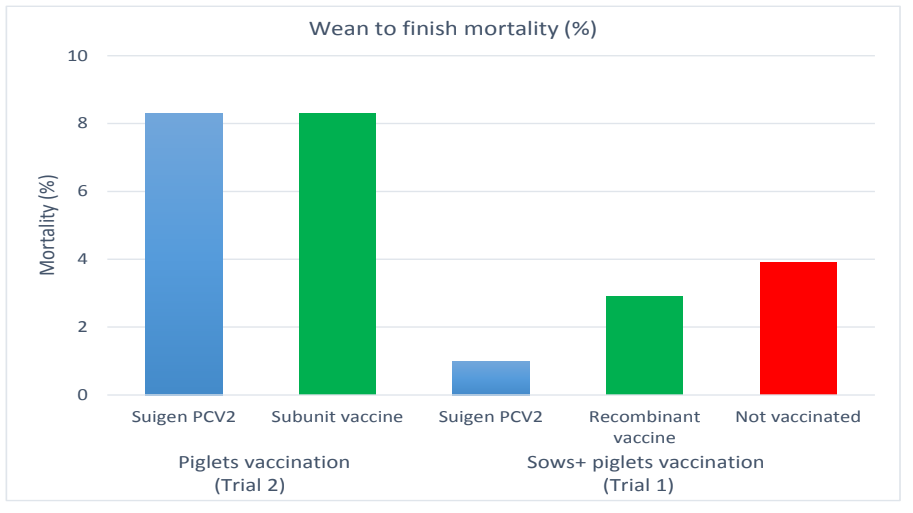
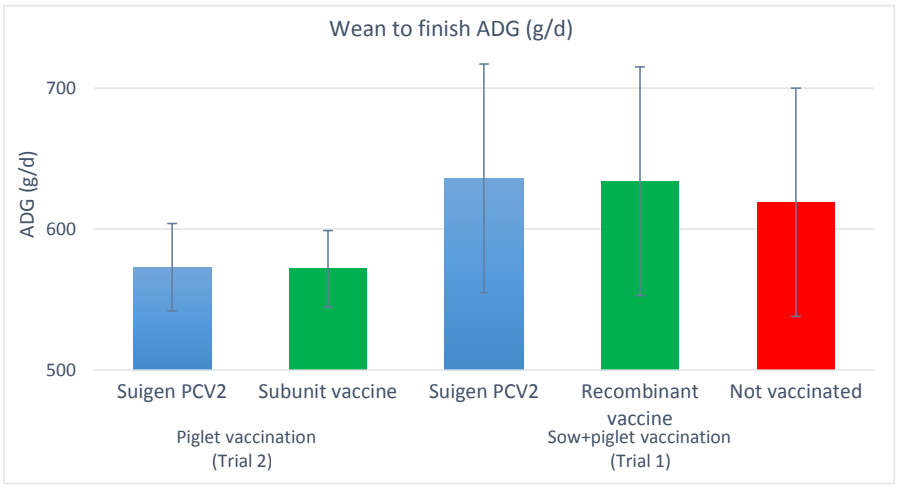
Trial 2 was performed in a farrow to finish farm owning 300 sows. One hundred twenty piglets were allocated to 2 groups of 60 each. Piglets were vaccinated against PCV2 around 6 weeks of age by the tested vaccine in group T (Suigen® PCV2, Virbac) and by the usual PCV2 vaccine of the farm in group C (subunit vaccine: 1 ml by IM route in both groups). Pigs were individually weighed at birth, weaning and finishing in both trials.
Local and general reactions were checked after vaccination in both trials as well as mortality cases till finishing. Categorical data and quantitative data were compared between groups by Fisher’s exact test and GLM respectively.